1. The electrical conductivity of aluminum or aluminum alloys is highly dependent on alloy composition and temperature. Generally, the lower the temperature, the higher the conductivity; the more alloying elements present, the lower the conductivity and the higher the resistivity.
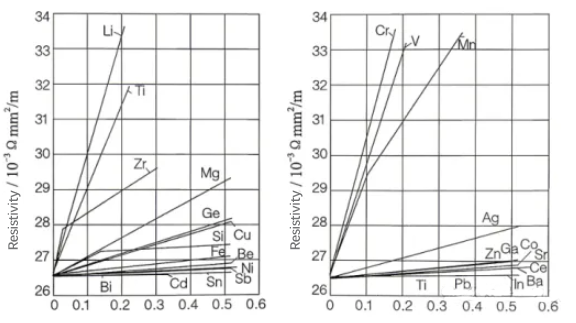
2. Figure shows the effect of high-purity aluminum with the addition of other elements to form binary alloys (annealed state) on the resistivity of aluminum. Among them, elements such as lithium, zirconium, titanium, chromium, vanadium, and manganese can cause an increase in resistivity. That is, small additions or contamination of these elements have a considerably adverse effect on the conductivity of aluminum. Lower conductivity also implies poorer thermal dissipation.
Relationship between element composition and resistivity for binary alloys based on pure aluminum.
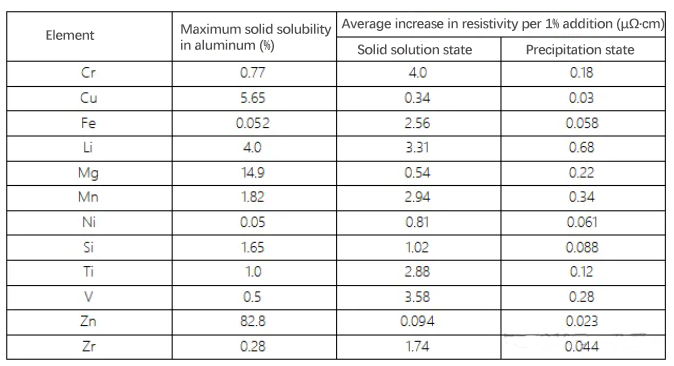
3. Some elements have low solid solubility and appear in the precipitated state. Generally, the solid solution state causes a more significant increase in resistivity than the precipitated state. The table shows the effect of alloying elements in aluminum in the solid solution state and the precipitated state on resistivity. The table indicates that the average increase in resistivity per 1% addition of an element in the solid solution state is significantly higher than that in the non-solid solution state.
Effect of each 1% addition of an element in aluminum on the average increase in resistivity – solid solution state vs. non-solid solution state.
4. Because the solid solution state results in even lower conductivity than the precipitated state, the conductivity of the alloy can be used to evaluate the quality of castings and products. For example, it can be used to investigate the precipitation of high-temperature dispersoids and the solid-solution/precipitation mechanism of strengthening phases in round bars, square ingots, semi-finished products, and finished products.
5. Some alloys contain chromium, manganese, vanadium, or zirconium. One purpose of homogenization heat treatment is to cause these elements to transform from the as-cast solid solution state to the precipitated state. Therefore, the conductivity of the alloy is measured to understand the dispersion of the dispersoids. In addition, conductivity is often used to investigate or verify the precipitation state during aging heat treatment.
6. Thus, using conductivity to predict and assess precipitates (including dispersoids and strengthening phases) in aluminum alloys is currently the simplest and most practical method.
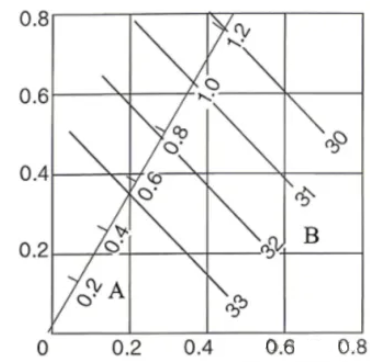
7. When three or more elements are added and form a solid solution state, the resistivity may exhibit an additive effect. However, once precipitates appear, the additive calculation of the effect of elements on resistivity is affected. For example, the conductivity of Al-Mg-Si alloys is related to both precipitation conditions and alloying elements. When the solid solution and precipitation conditions are fixed for an Al-Mg-Si alloy, conductivity is related to the Mg2Si content. As shown in Figure 2, the higher the Mg2Si content, the lower the conductivity; in other words, the higher the silicon and magnesium contents, the lower the conductivity. For fixed silicon and magnesium compositions, the conductivity of Al-Mg-Si alloys is lower in the T4 state because T4 involves the precipitation of GP zones (coherent transition zones of Mg2Si). The larger the precipitate size, the higher the conductivity. Hence, the conductivity of the T6 state is higher than that of T4, the T7 state is higher than T6, and the annealed state (O) has the highest conductivity.
Relationship between magnesium and silicon composition and electrical conductivity in Al-Si-Mg aluminum alloys.
X-axis: silicon content (mass fraction / %), Y-axis: magnesium content (mass fraction / %). Solution treated at 540°C, aged at 160°C for 16 h.
A – Mg2Si content (mass fraction / %), B – Conductivity (m/Ω·mm2).
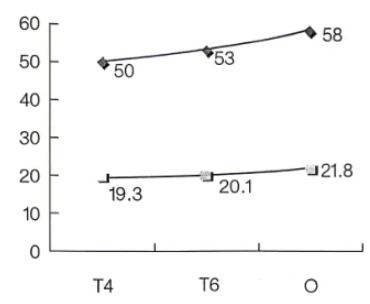
8. The figure below shows the conductivity and thermal conductivity of 6063 alloy in the T4, T6, and O states. For 6063-O, the average electrical conductivity is about 58% IACS; for T6, about 53% IACS; for T4, about 50% IACS. Thermal conductivity is proportional to electrical conductivity.
Relationship of heat treatment precipitation state with electrical conductivity and thermal conductivity for 6063.
(Top) Electrical conductivity (% IACS), (Bottom) Thermal conductivity (10 W/(m·K)).
9. Cold working has a slightly adverse effect on the conductivity of pure aluminum, but the effect is very small. As shown in the figure below, cold working causes a slight decrease in conductivity; the greater the deformation, the lower the conductivity.
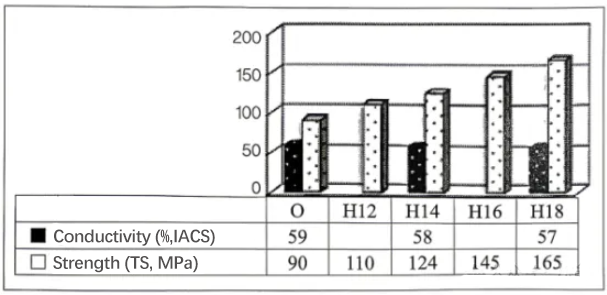
Relationship between cold working state, work hardening, and electrical conductivity for 1100 aluminum.
10.For 1100-O, the average electrical conductivity is about 59% IACS, and the tensile strength is about 90 MPa. Compared with the O state, H14 (about 35% cold reduction) has a tensile strength increased to about 124 MPa, but the average electrical conductivity is about 58% IACS, a reduction of only about 1% IACS.
11.Similarly, compared with the O state, 1100-H18 (about 75% cold reduction) has a tensile strength increased to about 165 MPa, but the average electrical conductivity is about 57% IACS, a reduction of about 2% IACS. Thus, cold working causes a slight decrease in conductivity.
12.By combining heat treatment and cold working, an appropriate microstructure can be obtained, enabling the alloy to achieve satisfactory mechanical properties and electrical conductivity.
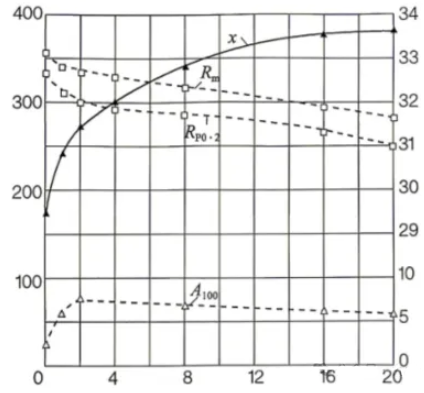
13.The figure below shows the relationship between aging time at 160°C and the mechanical properties and electrical conductivity of a conductive Al-Mg-Si (6101) alloy wire, which was solution treated at 525°C, naturally aged for 14 days, cold worked 95%, and then artificially aged. The figure shows that as aging time increases, electrical conductivity increases significantly, while mechanical strength or elongation decreases only slightly. In the 6101-T4 state, after 95% cold working, the tensile and yield strengths reach their maximum – tensile strength up to 355 MPa, yield strength up to 330 MPa, but elongation is only 2.5%. The electrical conductivity in this state is about 50% IACS.
Relationship of aging time at 160°C with mechanical properties and electrical conductivity for conductive Al-Mg-Si (6101) alloy wire. Solution treated at 525°C, naturally aged 14 days, cold worked 95%, then artificially aged. Rm – Tensile strength, Rp0.2 – Yield strength (MPa), Conductivity – k·m/(Omega·mm2), A100 – Elongation (%). X-axis: aging time.
14.After artificial aging at 160°C for 2 h, elongation increases to 7.5%. With further aging, elongation gradually decreases after 2 h; tensile and yield strengths slowly decrease, while electrical conductivity increases with aging time. This is due to precipitate aging and over-aging. Therefore, the aging treatment conditions must be determined based on the required electrical conductivity and strength of the material.
Post time: May-02-2026